Thus, Fe 2 is called the iron (II) ion, while Fe 3 is called the iron (III) ion. In the simpler, more modern approach, called the Stock system, an ion’s positive charge is indicated by a roman numeral in parentheses after the element name, followed by the word ion. The only other elements which form monatomic anions under normal circumstances are hydrogen (which forms H –ions) and nitrogen (which forms N 3– ions). There are two ways to make this distinction. Such nonmetals are found mainly in periodic groups 16 and 17. The number of nonmetals with which a group 1, 2, 3, or lanthanoid metal can combine to form a binary ionic compound is even more limited than the number of appropriate metals. All of the lightest colors are found on the left side of the periodic table and the darkest colors are found among the noble gas group. The general trend observed here is that the purple gets darker across a period. (CC-SA_BY-NC 3.0 anonymous) The periodic table is colored with different shades of purple. To name a binary ionic compound, identify the ions and list the cation first followed by the anion. Hydrates contain a specific number of water molecules attached to them.\) The image above shows the ionization energy of elements, with darker shades representing higher ionization energies. Here is the table with the names and formulas of polyatomic ions:īaSO 3 – Barium sulfite, Na 2CrO 4 – Sodium chromate, KClO 4 – Potassium perchlorateĮxamples of compounds containing a transition metal with two possible charges:ĬuNO 3 – Copper(I) nitrate or Cuprous nitrateĬu(NO 3) 2 – Copper(II) nitrate or Cupric nitrateĭon’t forget to add the prefix hypo– (meaning “less than”) and per– ( “more than”) for oxyanions with more than two formulas. The same principle of listing the names of the cation followed by the name of the anion applies to naming ionic compounds with polyatomic ions. The names and formulas of metals forming different cations are summarized in the table below:įeCl 3 – Iron(III) chloride or Ferric chlorideįeCl 2 – Iron(II) chloride or Ferrous chloride Binary molecular ( covalent) compounds are formed as the result of a reaction between two nonmetals. compounds are formed when a metal combines with a nonmetal to form a binary ionic compound. The suffix for the cation with a higher charge is “ ic” and for the lower charge is “ ous”. Name binary ionic and nonionic compounds. 
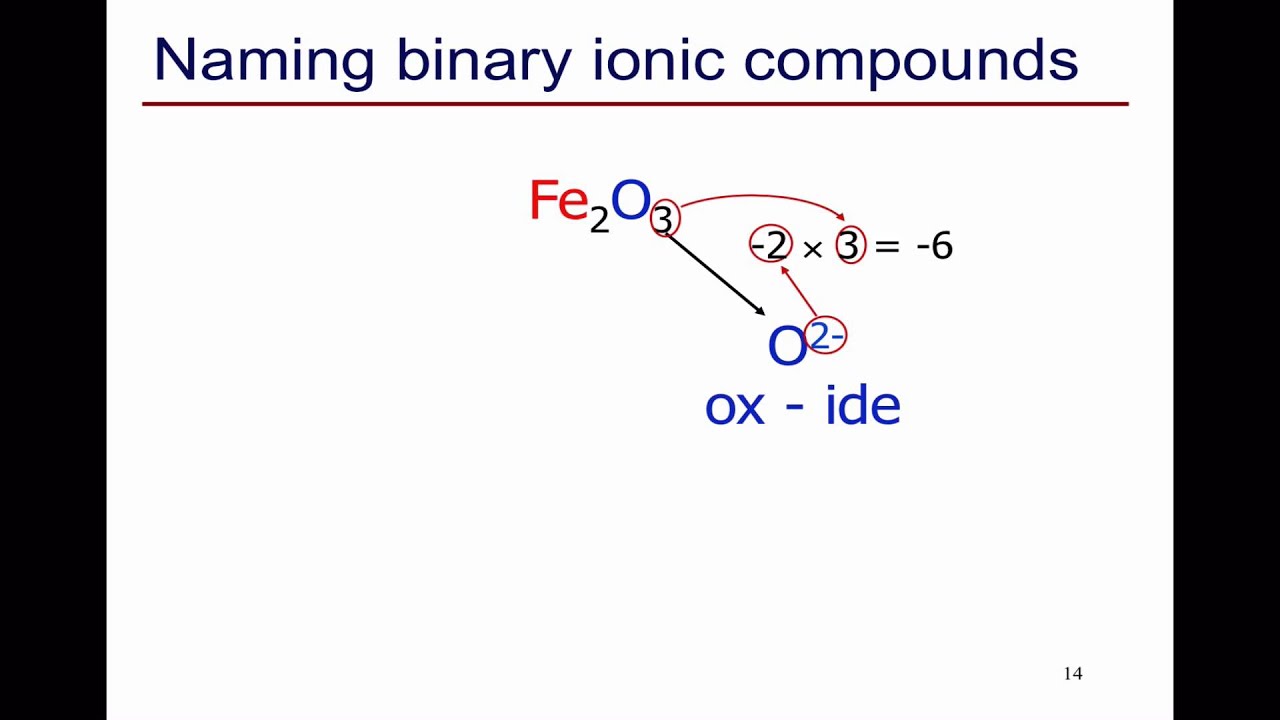
If the compound contains a cation of a transition metal with two possible charges, then a Roman numeral or the corresponding suffix is used to indicate its charge. There are two ways to make this distinction. K 2S – Potassium sulf ide, CaBr 2 – Calcium brom ide, AlCl 3 – Aluminum chlor ide, MgO – Magnesium ox ide Remember, group 1 and 2 cations inherit the name of the metal they are derived from, and monoatomic anions take the ending –ide. Binary Compounds - They are the compounds formed from the metal cation and the nonmetallic anion, such as sodium chloride - NaCl. Here both elements are ions (an anion which has a negative charge and a cation which has a positive charge). To name a binary ionic compound, identify the ions and list the cation first followed by the anion. Binary ionic compounds are salts which consist of only 2 elements. Once you know the names of the common ions, it is not difficult to name ionic compounds as we only need to put the names of the cation and the anion together.īinary ionic compounds are made of only two elements. The name of a binary ionic compound is: metal nonmetal -ide If the metal has different oxidation states, the oxidation state is named. In the previous post, we talked about naming monatomic (monoatomic) and polyatomic ions. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
